What is the pH Balance of the Body?
The pH level is one of the most
important balance systems of the body. The term
pH stands for “potential” of
“Hydrogen”. It is the amount of
hydrogen ions in a particular solution. The more
ions, the more acidic the solution. The
fewer ions the more alkaline (base) the
solution. The pH level is a measure of acidity or
alkalinity, on a scale of zero to fourteen, with zero being most acid,
fourteen being most alkaline and seven being mid-range. The most critical
pH balance is in the blood.
|
 |
|
Acidic Alkaline |
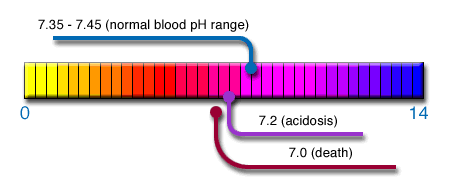
Normal blood pH has a very
small window of acid/alkaline pH balance.
Blood pH must range between 7.35 and 7.45. This
means that there is an adequate amount of
oxygen in the blood. Any
slight decrease in pH will result in lower
oxygen levels in the blood and, therefore, in
the cells. Any drop in pH, no
matter how slight, is the beginning of a disease
state and affects when and how we age.
All other organs and fluids will
fluctuate in their range in order to keep the blood
at a strict pH between 7.35 and 7.45 (slightly
alkaline). This process is called homeostasis. The
body makes constant adjustments in tissue and fluid
pH to maintain this very narrow pH range in the
blood. A normal pH
of all tissues and fluids of the body (except
the stomach) is slightly alkaline. The stomach pH is
much more acid than the intestinal pH because
the stomach needs an acid environment
(hydrochloric acid) to break down food for
digestion. Whereas, the flora (good bacteria) of
the intestine need a more alkaline environment
to assimilate and process the nutrients from the
foods digested by the stomach.
How
Does Eating Affect Our pH Level?
|
Foods for a Balanced pH
For a naturally balanced pH,
avoid over consumption of
the following:
- meat
- alcohol
- soft drinks
- caffeine
- coffee
- most nuts
- eggs
- vinegar
- sauerkraut
- ascorbic
acid
- cheese
- white sugar
-
medical drugs.
Add more of these to your diet.
- RAW vegetables
-
ripe fruits
-
bean sprouts
-
CLEAN water
-
milk
(goat's is best)
-
onions
-
figs
-
carrots
-
beets
-
miso
|
|
Diet is probably the most
important change we can make to balance our
natural pH. We need to eat at least 75% alkaline-forming foods. The
average all-American diet consists of about 80% acid-forming foods!
Because processed and refined foods are extremely acidic to our systems,
the body creates a buffering system (a chemical process to protect the
body from being harmed by the acids). This buffering process requires
the use of many nutrients from the body, including electrolyte minerals
(organic potassium, magnesium, sodium, calcium, to name a few).
Electrolyte minerals are not minerals from the ground. They're minerals
from plant sources that have gone through the process of photosynthesis.
Why are Electrolyte Minerals Important
for our Bodies?
Electrolyte minerals are vital to the
metabolic functions of our body systems. When we have a sufficient
reserve of electrolyte minerals, the buffering process (the cushioning
and removing of unwanted acids from our systems), is not a problem. When
we are young, we usually have sufficient reserves. Over time, the
electrolyte mineral reserves can become depleted by ingesting too many
processed and refined acid-forming foods, or from excessive mental or
emotional stress. When we run short of electrolyte minerals, our body is
no longer able to maintain an efficient homeostasis (a state of
equilibrium).
The body has a hierarchy of priorities
for survival. Second only to breathing and sustaining our heartbeat, the
most important metabolic function that our bodies perform is maintaining
a specific pH. The most important pH level that the body must regulate
is the blood’s pH level. The body’s blood pH level must be maintained at
7.4 (slightly alkaline). If it varies more than a point, death can
result from a coma or a seizure. In order to maintain the blood’s
critical pH balance, the body will compromise less important functions.
Once the electrolyte reserves become depleted, the body begins to rob
these electrolytes from the various organs and systems of the body to
maintain the blood’s pH level. This is where the imbalance begins.
The
following are examples of altered pH levels and the resulting imbalanced
internal body system:
If the bowels and intestines are robbed
of electrolyte minerals, the pH level becomes altered. This creates an
imbalance in the bacterial environment, which can leave the bowels and
intestines open to pathogens (including candida and parasites),
irritations and disturbances. Have you ever heard of irritable bowel
syndrome or leaky gut syndrome?
Also, when the body becomes depleted of
organic sodium (an essential electrolyte mineral, not table salt), the
body may not be able to manufacture enough hydrochloric acid in the
stomach. This can create an imbalanced digestive system, where foods are
not being digested properly.
These imbalances can be aggravated
further because our body systems will generally deplete electrolytes
from the weakest areas first. So if someone already has a weakened body
system, the further depletion of electrolyte minerals will cause a
greater imbalance and more dysfunction. When our bodies become too
depleted, overall functioning becomes weakened, affecting the immune
system and the body’s ability to fight off bacterial and viral
infections.
Testing
the pH Level
You can easily monitor your
pH with simple testing strips
which can be purchased at your
local pharmacy. Testing saliva is the most effective way to gauge
the body’s
pH. To test saliva: Wait 2 hours after eating.
Spit into a spoon. Dip the strip. Read immediately. Use the color chart from the correct indication. An
optimal reading is 7.5. This indicates a
very slightly
alkaline body.
To test urine: Test a urine sample first thing in
the morning. Fill a small cup with urine, and dip a
strip into the cup. Read immediately. Results: 7.0
is neutral. A reading of 6.5 is slightly acidic. A
reading below 6.5 is very acidic. Note: A reading of
8.0 or above, while common, indicates a body that is
too alkaline. Urine is slightly more acidic than
saliva. [from pH strip producer: Phion, Inc.]
About
Balancing the pH
There are many
good products out there for raising pH levels.
Your diet and water are most important, however.
Below are a few of our favorite ways for quickly
raising pH levels
and oxygenating
tissue.